To see how well you know the information, try the Quiz or Test activity. When you need a break, try one of the other activities listed below the flashcards like Matching, Snowman, or Hungry Bug.Īlthough it may feel like you're playing a game, your brain is still making more connections with the information to help you out. If you are logged in to your account, this website will remember which cards you know and don't know so that theyĪre in the same box the next time you log in. LEFT ARROW - move card to the Don't know pile.You can also use your keyboard to move the cards as follows: If you've accidentally put the card in the wrong box, just click on the card to take it out of the box. When you've placed seven or more cards in the Don't know box, click "retry" to try those cards again. If you knew the answer, click the green Know box. Look at the large card and try to recall what is on the other side. Use these flashcards to help memorize information. as energy level continues to fill it has a stronger attraction for the elecrons. INCREASES from left to right along a periodġ. Trends in Electron Affinity going left to right across a period How strongly an atom attracts additional electrons The amount of energy required to add an electron to a neutral atom to form a negative ion or anion. Reactivity increases moving up the group.ġ: Decreasing atomic radius brings the outer shell closer to the nucleus.Ģ: Electronegativity increases as we move up the group.ģ: All the halogens require only 1 electron to achieve noble gas structure This means they have a low ionisation energy and electronegativity.ġ: Easier to cut as we move down the group.Ģ: Display more vigorous reaction with water as we move down the group.ģ: Tarnish with air faster as we move down the group. 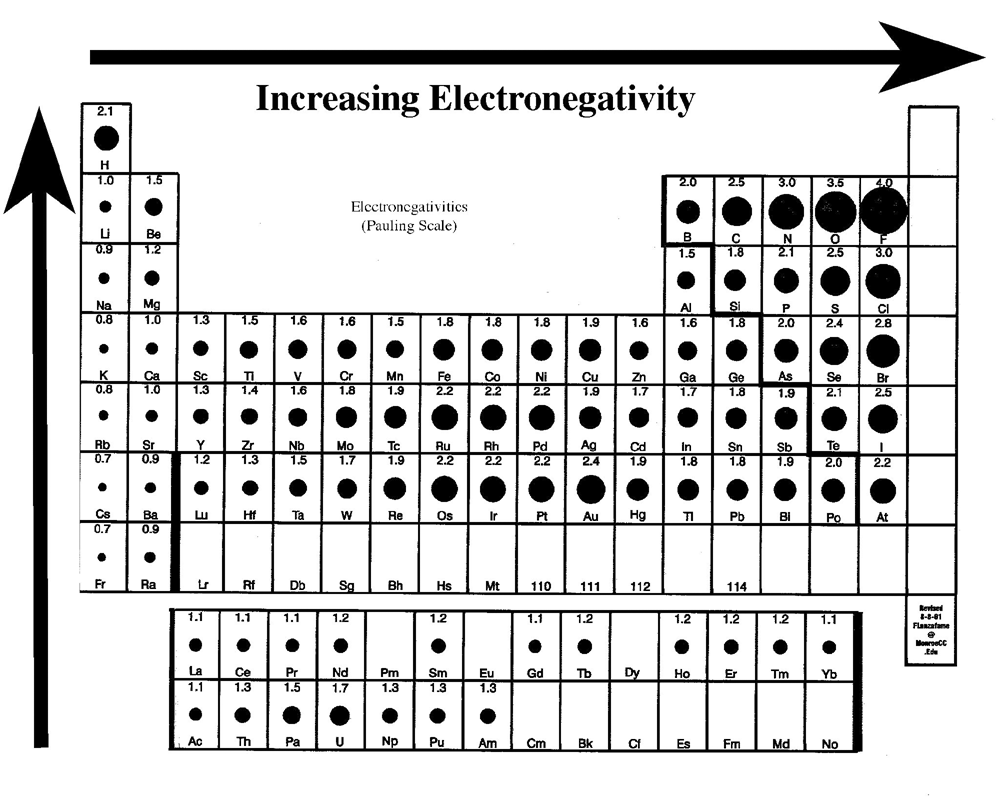
Losing this electron gives them a full outer shell. Ionization Energy INCREASE going across a periodġ: Increasing nuclear charge means the nucleus has a better hold on the outer electrons.Ģ: Decreasing atomic radius means that the outermost shell is closer to the nucleus and is therefore held tighter.Īll the elements have only 1 electron on their outer shell. 
Trends in Ionization energy going left to right across a period: 2 reasons Ionization Energy DECREASES going down a group.ġ: As atomic radius increases the electrons are further away from the nucleus.Ģ: The screening effect of the inner electrons cancels out the pull of the nucleus. Trends in Ionization energy going down a group: 2 reasons Noble Gases have the highest Ionization Energy levels, as they have full outer energy level The first ionization energy of at atom is the minimum energy required to completely remove the most loosely bound electron from a neutral atom in a gaseous state. 2 reasonsĮlectronegativity INCREASES going across a period.ġ: Increasing nuclear charge means the nucleus has a stronger hold on the electrons.Ģ: Decreasing atomic radius: as no new shell is being added the stronger nucleus can hold the outer electrons tighter Trends in Electronegativity going left to right across a period. 2 reasonsĮlectronegativity DECREASES as we go down a groupġ: Increasing atomic radius means the electrons are further away from the attractive force of the nucleus.Ģ: The screening effect of the inner negative electrons block the pull of the positive nucleus. Trends in Electronegativity going down a group. Linus Pauling developed the electronegativity scale. Will predict how atoms combine chemically. 2 reasonsġ: Increasing nuclear charge due to an extra proton being addedĢ: There is no increase in screening effect as no new shell is being addedĪ measure of an atom's ability to attract and hold electrons in a molecule. Trends in Atomic Radius going left to right across a period. 
2 reasonsġ: A new shell is being added as we move down the table.Ģ: The screening effect of the extra electrons cancels out the pull of the growing nucleus. Trends in Atomic Radius going down a group. Periodic Trends Trends and Patterns in the Periodic Table QuestionĪtomic radius is defined as half the distance between the nuclei of atoms of the same element that are joined by a single covalent bond.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
